Persistence of Long-Term Memory in Vitrified and Revived Simple Animals
See also the published paper: Persistence of Long-Term Memory in Vitrified and Revived C. elegans. Rejuvenation Research. (doi: 10.1089/ rej.2014.1636)

From Cryonics September 2015
By Natasha Vita-More, Ph.D.
“If the aging process is controlled in a similar way in worms and humans, then we can use what we learn about worms to speed our study of higher organisms.” — Cynthia Kenyon
Demonstrating the preservation of memory after cryopreservation is a crucial step for cryonics. The research leading to this breakthrough will help to build momentum toward more advanced studies on information storage within the brain, as well as short term behaviors of episodic, semantic, procedural, and working memory.
In this article, I will review how I became involved in this research, the guidance along the way, my initial training at 21st Century Medicine, pitching the research project to Alcor, and submitting my proposal to the Alcor Research Center (ARC). I will then take you into the lab, the process of trial and error in our first studies, developing a protocol based on olfactory imprinting and applying several cryopreservation methods, developing the migration index, and the rewards of working with a lab technician who became an admired colleague.
From this experience and looking toward the future, I am more committed than ever to support and help lead scientific research projects that enrich learning about memory after cryopreservation. But this does not come without the insight to imagine, to speculate, and to hypothesize. Observing a gap in the current state of things triggers a desire to understand why there is a gap and to do something about it. From there we can query until one idea sticks and garners enough value to move forward. For me, this one idea was all about memory retention.
The lingering concern: ‘How can something that cannot be demonstrated be scientific?’ found in the Alcor FAQ has now been addressed. There is some existing evidence of preservation of neural structures as demonstrated by electron microscope studies. Yet these studies observe static structures and not survival of memory in practice. While the larger question of how a person’s identity can be sustained after cryonics has not been conclusively answered, it is a fact that long-term memory is retained in a simple animal. This experience causes me to think back on Neil Armstrong’s statement after the Apollo 11 Mission. Certainly not as grand, but nevertheless, “This is one small step for a [nematode], but one giant leap for [cryonics].”
Developing the Research Project
This research was to put into motion as a project I had been musing about for many years that concerns the outstanding issue of cryonics and memory retention. While the science and technology of cryopreservation have advanced over the past decades, there had been no direct evidence that an animal could be cryopreserved, revived, and tested for memory retention with positive results.
During the 25 years I have been a member of Alcor, I have listened to the internal conversations among cryonicists and read public commentary about the viability of cryonics. A core question has been: Will you remember who you are if and when you are revived? While this question can only be answered definitively once the first cryopreserved person is revived, it seemed logical that there needed to be small, baby steps along the way. Several people had begun projects to explore this area, but none had been conclusive, let alone published.
As a bit of background, some of my colleagues in the field in which I pursued my doctorate had done interesting biodesign experiments. Dr. Edwardo Kac had developed the transgenic “GFP Bunny,” Stelarc succeeded in cloning and transplanting his ear onto his arm, and Dr. Ionat Zurr with Oron Catts had developed tissue culture as “semi-living” sculptures. Yet, there was an identifiable lack of exploration and experiments in the biodesign field of human enhancement and life extension that linked directly to cryonics.
Dr. Greg Fahy, a leading cryobiologist, had been an exceptional mentor since the inception of this project. He had told me about a researcher’s work that captured stunning visuals of human sperm as they absorbed glycerol and were obscured by ice formations, until the ice receded and they began moving again. Inspired by this, I set out to study what types of life forms I could work with and which exhibited unique physical movement. Based on Greg’s advice, I decided to work with C. elegans.
“Caenorhabditis elegans is one of the most important models used in biology and neurology and has countless applications in the area of biological sciences. The simplicity of its size (1mm), the transparency of its neuronal network (hermaphrodites contain 302 neurons), and its short but complex life cycle make C. elegans of potential value to studies of memory retention after cryopreservation.” (Vita-More & Barranco, 2014).
C. elegans can be trained through nonassociative learning, associative learning, and imprinting. They can habituate to chemical stimuli and learn smells, tastes, temperatures and oxygen levels. They also respond to vibrations, such as tapping on the petri dish. In regards to cryonics, C. elegans have high survival rates, with little to no cryoprotectant, when using ultra-rapid cooling and warming methods. By providing a case where I could use a viable learning environment for the worms, cryopreserve them with their structure intact, revive them, and then test their memory of the learning behavior, I might be able to add significant research to the field of cryonics. I spent the next year or so looking for grant money to support the research. Eventually persistence paid off, and Fahy was consequential in my obtaining the grant from Alcor Life Extension Foundation.
“Memory models that are amenable to testing after cryopreservation are not plentiful. The best test of memory is behavioral, but there are no easily accessible organisms more complicated than C. elegans that can be cryopreserved whole to enable behavioral tests after rewarming. So I think Natasha’s proposal is appropriate for pushing the envelope given the constraints involved. Perhaps success in this project could serve as a jumping off point to testing polar insects or Siberian salamanders down the line, but first things first. You have to walk before you can fly.” (Fahy, 2013)
The question I asked in this research was whether memory could be retained after cryopreservation. This single question became the object of the research. To attempt to answer this question, C. elegans was the model organism for testing. It is a known model used in biology and neurology, with the simplicity of its small size, and it had already been successfully vitrified and (separately) trained. But there had been no research experiments combining both vitrification and cryopreservation, and also training and testing memory after reviving. In short, it was the only simple animal where cryopreservation and revival had been demonstrated and a well-defined assay of learning had been completed.
Starting with the completed research performed in these two areas, my team sought to build upon these experiments in our study titled “Persistence of long-Term Memory in Vitrified and Revived C. elegans.”
Setting up the Lab at Alcor

After receiving the grant to begin the research, the Alcor team worked with me to locate a work area with space for a hood, and then I started ordering supplies. Biochemist Hugh Hixon advised me about basic chemistry and we selected an aluminum mini-Dewar for holding the liquid nitrogen for the vitrification studies. We prototyped several methods for detecting worm migration on slides and petri dishes. Technology innovator Steve Graber created the lab area, set up the hood, and worked with me to test microscopes for depth of field, lens magnification, and our video recording of the studies. Dr. Mike Perry worked with me to brainstorm statistical software for analyzing the migration rates of tested worms.

Task chart to keep track of things in the lab.
Fahy had introduced me to Dr. Ramon Risco, founder of the CryoBio Tech Research Group. He provided the protocol for a particular method of vitrification, known as the slush method. This method uses quartz capillary tubes that have a specific diameter and require a slush making apparatus. Hugh ran with this idea and built our slush system.

Steve, Hugh and Mike testing Hugh’s slush system.
We were excited to be moving forward with the research and making some progress; nevertheless, one core issue was that I needed another researcher and a highly skilled lab technician to work directly with me in the lab on a day-to-day basis. I contacted Chris Rasch, who had worked with cryopreservation of organs and had knowledge about C. elegans. He helped in developing preliminary learning protocols for training the worms, such as tapping on petri dishes which elicits a backing reflex, using lighting stimulation which evokes an avoidance response, and introducing chemical attractants such as Butanone for Pavlovian conditioning.
Risco had introduced me to Daniel Barranco, a current Ph.D. candidate, who is an expert in the Cryotop method of embryo freezing. We had been communicating for many months about various supplies and chemicals. But since Barranco lives in Spain and the phone calls and Skype meetings were becoming lengthy, I invited him to work with me in the lab. His strong skill set was a key factor in our iterative process of exploring options for learning methods, cryopreservation protocols, and testing, retesting, and finally determining a process for accurately assessing the long-term memory retention of the cryopreserved and vitrified worm studies.



Natasha in the lab; Daniel at the hood; Natasha vitrifying worm.
Validation of Memory Retention in Studies
The memory retention protocol we used for learning is known as olfactory imprinting. We distinguished this protocol by using the chemical benzaldehyde for phase-sense imprinting on the young worms, just after the larval stage. Olfactory imprinting has been studied in many species, including primates, mammals and humans. The key to successful olfactory imprinting is choosing the correct period of time (window of opportunity) when the organism can develop a long lasting learned response.
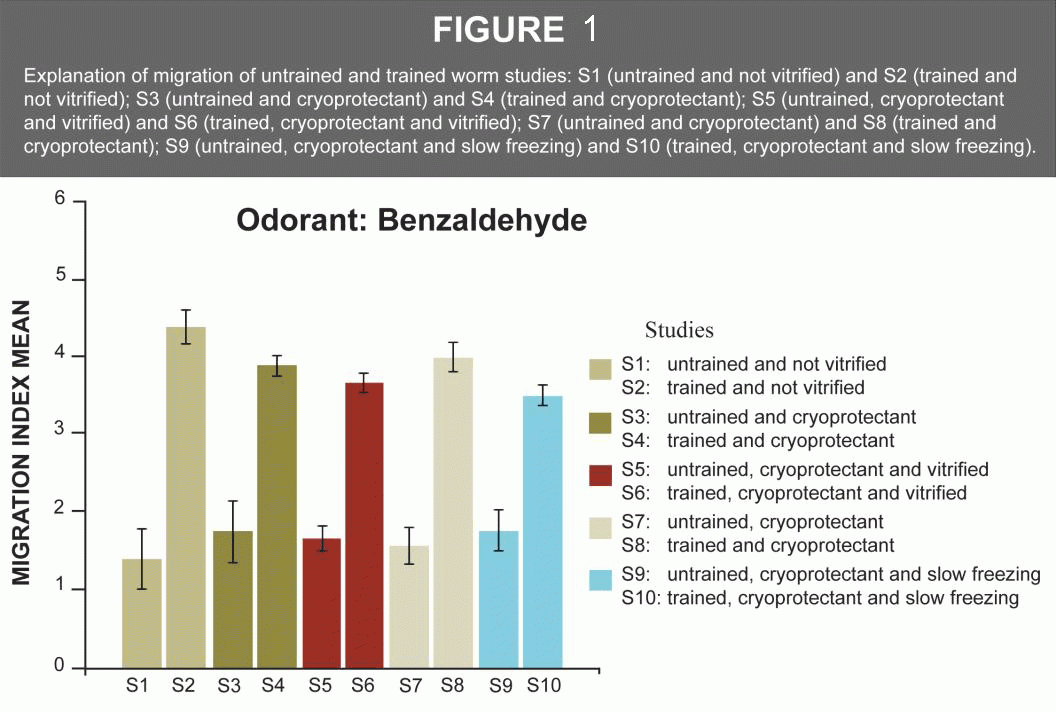
Memory retention was validated through a chemotaxis assay of the migration index. The trained worms migrated to areas of the petri dish where the benzaldehyde drops were placed. This showed that they preferred areas of the dish where the chemical smell was detected. Because there is a native reaction to benzaldehyde, the untrained worms preferred other areas of the dish. In sum, the response of the trained worms was double that of the untrained worms, whether they were cryopreserved or not.

X marks the spot where worms migrated on the plates.
Establishing the Control Group and Experimental Group for 10 Studies
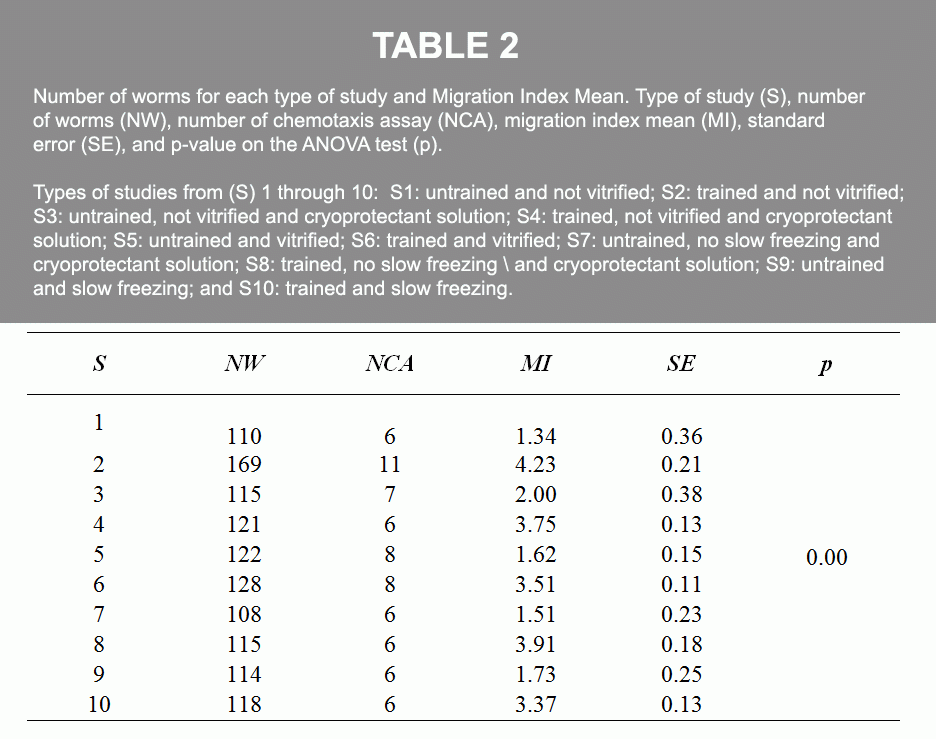
The research established two groups, the control group and the experimental group. For the control group, we formed eight studies. For the experimental group, we formed two studies. Each of the ten studies from the control group and experimental group contained 100 or more worms each (Table 1). We used the Wild isolate Bristol strain N2 of the C. elegans worm, which we obtained from the Caenorhabditis Genetics Center (CGC) at the University of Minnesota.
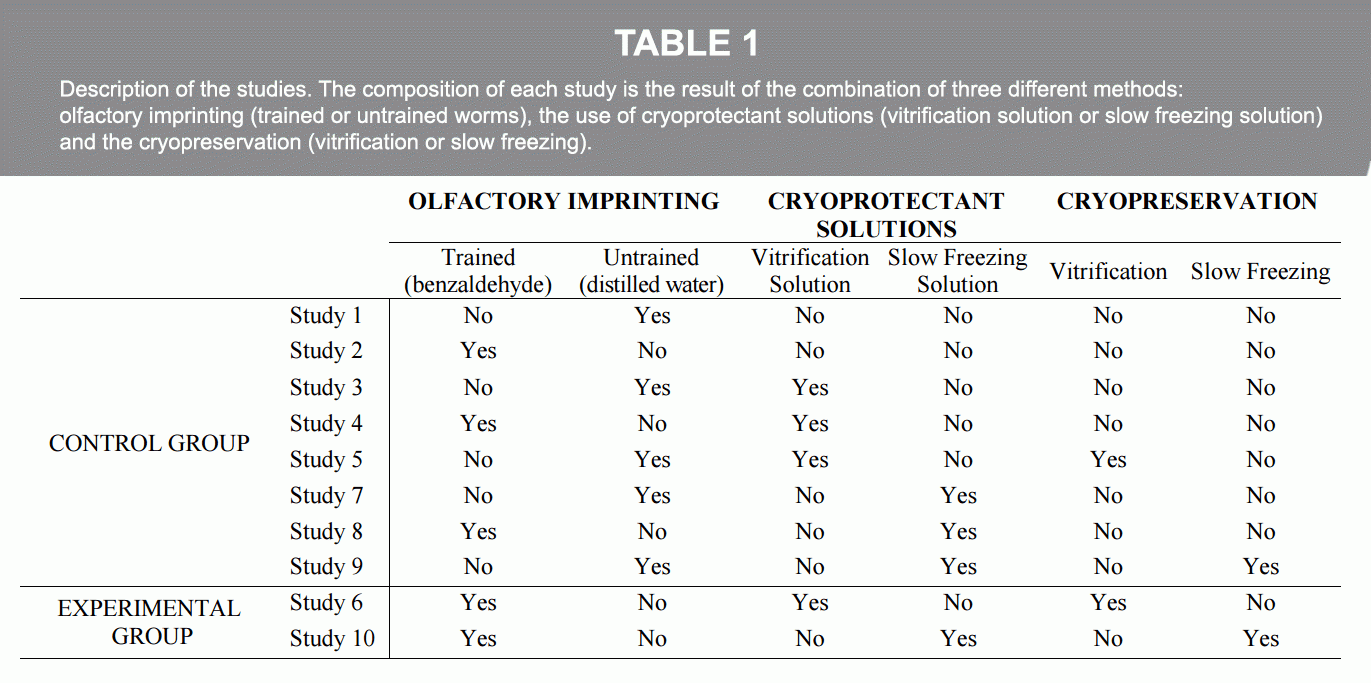
Methodology: Three Areas of Focus
Our methodology was based on what was already known in the field and what might be the most effective tools and techniques to use. After much deliberation, we decided to incorporate an established method for learning, several methods for cryopreservation, and a chemotaxis assay for observing whether or not the worms had remembered what they learned at the early L1 stage and after cryopreservation and reviving at the adult stage.
1. Learning Method: Using the olfactory imprinting method (Remy and Hobert), our studies focused on long-term memory using this olfactory imprinting technique with the chemical attractant benzaldehyde (C6H5CHO). The C. elegans habituate to learned smells associated with food and the best results are at an early stage, just after the worm develops from the larva stage. This is the opportune period of time for the animal to develop a longlasting learned response.
In our experiment, we used 100 or more worms for each study and used the term untrained for worms that we did not imprint with benzaldehyde. For worms that we did expose to the benzaldehyde for olfactory imprinting, we used the term trained. Using the protocol of Remy and Hobert, we placed worms in the untrained group in petri dishes where food Escherichia coli (E. coli) was available and the dish lids were swiped with distilled water. For the trained group studies, we placed the worms in petri dishes with E.coli where the lids of the dishes were swiped with benzaldehyde, every hour for eight hours.

Swiping lid with benzaldehyde.
In the studies, the benzaldehyde was used as an attractant, which developed an association between food and the chemical smell. The aim was to establish whether or not the nematodes could retain the imprinted experience of the chemical smell of benzaldehyde with food into their adult stage, signifying the establishment of longterm memory.
2. Vitrification and Cryopreservation Process: The traditional methods for cryopreserving biological samples are through slow freezing and through vitrification, which have different cooling and warming rates. For our research’s vitrification, we applied the known method of Cryotop, used in the freezing of embryos. While our research experiment’s studies included several methods for cryopreservation, our central focus was the Cryotop protocol indirectly submerging the nematodes into liquid nitrogen using a straw device. One worm at a time was carefully pulled into the straw from the petri dish. From this, we established the effective use of the SafeSpeed closed device (Barranco, et al.), a new technology for ultra-fast warming rates.


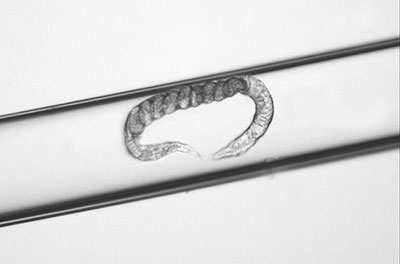
Pulling worm into straw.
3. Testing Results of Long-Term Memory. We used a chemotaxis assay five days after olfactory imprinting, when worms reached the adult stage. Marking 12×12 square agar plates, we drew lines marking off areas with assigned values of from -6 to 6 on the outside of the plates. In the first area of the plates, at value -6, we issued three drops of sodium azide at equal spacing into the agar. In the same areas, with the same equal spacing but on the lid of the plates, we issued three drops of plain water. On the other side of the plate, at value 6, we issued the same three drops of sodium azide at equal spacing; but on the lid of this area, we issued three drops of benzaldehyde, instead of water.

Pipetting three drops sodium azide.

Pipetting three drops benzaldehyde.
A series of processes included using a platinum wire to pick up revived worms from the petri dish with food, to a petri dish without food, and after numerous minutes, transfer them onto the square plate to time and observe where they migrated to. This was the Migration Index (MI). The statistical analysis for each study was tested with the Levene test, ANOVA test, and Tahame test (Figure 1 and Table 2).
A Brief Description of the Research Results
The research shows the first results related to persistence of long-term memory of C. elegans after vitrification and reviving. I, along with Daniel Barranco, describe the results in our paper in Rejuvenation Research (October 2015 issue):
Conclusion: This study introduced the C. elegans nematode to olfactory imprinting for learning with the specific goal of testing the animal’s memory retention after cryopreservation and revival. Our results show that the mechanisms that regulate odorant imprinting (a form of long-term memory) in young C. elegans have not been modified by either the process of vitrification or by slow freezing in the adult stage. This is the first evidence of preservation of memory after cryopreservation (vitrification or slow freezing).

Vitrified and revived worm in orange food coloring (Barranco, 2014).
A necessary future for researching memory retention of cryonics patients
The Alcor Research Center work has been a wonderful opportunity to build a project related to cryonics. I would like to lead a team or advise a team. As for extending C. elegans research, I would like to explore alternative learning methods at different maturity stages of the worm. Also, more work is needed to find out if a few or all memory mechanisms are unaffected by the Benzaldehyde and/or vitrification. Beyond this, I am interested in testing memory of larger organisms with a more complex central nervous system and leave others to continue the research that I and Barranco completed.
If I could do similar research in an animal with a more complex nervous system, I might consider cold-tolerant species that live suspended in a frozen state during winter seasons and thaw in the warmer seasons. The Greenland Woolly Bear Caterpillar is a species that is active for a mere 30 days of the full 365 days in a year, and then goes dormant in self-made cocoons. These cocoons are cleverly attached to rocks and the cocoon coverings form tiny biosphere greenhouses. Another species is the Alaskan Wood Frog, an amphibian that freezes solid through the winter and defrosts in the spring.
Nevertheless, after working with C. elegans, which naturally have rhythmic movements that are visually pleasing and emotionally alluring, it would be difficult to work with a leech, which is another option. The ozobranchid leech, a parasite that attaches itself to freshwater turtles, is a highly tolerant organism to freezing conditions and thawing, repeatedly. The downside is that these leeches can carry viruses that form cauliflower-like tumors on the turtles, impairing their health and survival rate. So here is a note of caution; however, they are known not to affect humans.
General Reflections on the Research
In the first few weeks, there were over 16,000 downloads of our paper, which is published ahead of print at Rejuvenation Research’s online site, http://online.liebertpub.com/doi/10.1089/rej.2014.1636. I would have been delighted if 600 people downloaded our paper from the publisher’s site. Writing the paper was not an easy task. It took considerable attempts to adjust the language and formatting, which is quite different from a doctoral dissertation in style. The critical and highly valued blind peer-reviewed comments were enormously helpful. And persistence paid off once again, and the paper was printed in the October issue of Rejuvenation Research.
The next step is to take the array of video footage and edit it into a visual documentation of the research and a stunning graphic narrative. Our continuous recording of the research provides sufficient visualization of the process, as well as distinct instances showing where the worms migrated. The aim is to capture the research studies with visuals, similar to the rhythmic movement of the C. elegans as they skim to and fro across the agar in the petri dish.
An Unexpected Incident
An unexpected incident occurred after I had vitrified a worm and took it from the warming bath to the petri dish with agar. I was carefully watching it to check on its behavioral movements. Looking through the microscope, I noticed some bubbles, or what I thought were air bubbles I had mistakenly emitted from the straw. But as I began to notice, four bubble-like shapes were not air bubbles. They started moving. Over the next minute or so, all four larvae had hatched and were healthy looking new baby worms. The vitrified and revived worm’s eggs were hatching before our eyes. This was one of the most thrilling moments.

C. elegans lays four eggs after vitrification and reviving. (Vita-More 2014).
REFERENCES:
Vita-More, N, Barranco, D. Persistence of Long-Term Memory in Vitrified and Revived C. elegans. Rejuvenation Research. (doi: 10.1089/ rej.2014.1636).
Brenner S. The genetics of Caenorhabditis elegans. Genetics. 1974;77(1):71-94.
Barranco D, Cabo V, Corral A, Risco R. Vitrification of Caenorhabditis elegans by ultrafast warming rates. Nature Methods (in preparation). 2014.
Fahy GM, MacFarlane DR, Angell CA, Meryman HT. Vitrification as an approach to cryopreservation. Cryobiology. 1984;21(4):407-426.
Fahy GM, Wowk B, Pagotan R, et al. Physical and biological aspects of renal vitrification. Organogenesis. 2009;5(3):167-175.
Remy JJ, Hobert O. An interneuronal chemoreceptor required for olfactory imprinting in C. elegans. Science. 2005;309(5735):787-790.
About The Author

Dr. Natasha Vita-More is a full time Faculty member and the Graduate Studies Program Champion (Chair) at the University of Advancing Technology. She earned her Ph.D. in Emerging Technology and Design at the University of Plymouth, UK, and a Master of Science from the University of Houston. Her doctoral dissertation outlines the study of life expansion as a framework for design-based approaches concerned with prolonging human life and sustaining personal identity. Natasha is Chair of Humanity+ and a Fellow at the Institute for Ethics and Emerging Technologies. She has been an invited speaker on human enhancement at the International SportAccord Convention for Olympic and non-Olympic sports, the TOPOS Conference in Tokyo on the world’s aging population, Russia’s Geek Picnic, and at Berlin’s CityCube. She has been published in numerous academic, scientific, and design journals. She has been called an “early adopter of revolutionary changes” and a “role model for superlongevity” (Wired magazine), and has appeared in over twenty-four televised documentaries on the future. She lectured at Harvard, Yale, Stanford, Virginia Commonwealth, Aalto (Finland), and Polytechnic (Hong Kong) universities. Her book, The Transhumanist Reader: Classical and Contemporary Essays on the Science, Technology, and Philosophy of the Human Future (Eds. More, M. and Vita-More, N, 2012), published by Wiley-Blackwell, is a foundational collection of ideas and essays by 42 authors on the human future.
Image: Steve Graber, 2014.
