Histological Study of a Temporarily Cryopreserved Human
From Cryonics, November 1984
HISTOLOGICAL STUDY OF A TEMPORARILY CRYOPRESERVED HUMAN
The case history of the present subject, referred to as P3 elsewhere (1), has been previously described [Postmortem Examination of Three Cryonic Suspension Patients]. Essentially, the patient was cryopreserved a significant time after clinical death and was stored for several years in liquid nitrogen until it became necessary to terminate cryopreservation of most of the patient’s body. After thawing of the non-cephalic portion of the body, tissue samples were taken and were preserved in either Karnovsky’s fixative (1) or in buffered formalin fixative (1) and processed some time later for standard light microscopic observation. The results represent the first direct information ever obtained concerning the effects of cryopreservation, as carried out under real working conditions, on the cellular and noncellular structural integrity of the human body. Although the conclusions which can be drawn from this single case are limited in several ways as will be described in the discussion which will follow, the results are such as to provide considerable encouragement for those individuals who now are or who someday will be considering cryopreservation as a personal alternative to death.
MATERIALS AND METHODS
Removal of Cryoprotectant from Tissue Samples. All tissues were fixed as described elsewhere (1) on January 4, 1984. The tissues were then stored at room temperature until March 19, 1984. On this date the tissue samples were decanted, blotted, and weighed and the volume of fixative used to store each tissue sample to this point in time was similarly measured. The results are shown in Table 1. In all cases, the volume of fixative used was more than 10 times the weight of the tissue sample. At this time, the liver samples were floating on their respective fixatives and appeared radically abnormal in color (greenish white). The fixatives for these samples were also quite turbid, in contrast to the transparent appearance of the fixatives bathing the remaining samples.
Table 1: Sample Preparation for Histological Observation
| 1° fixative* |
K
|
F
|
K
|
F
|
K
|
F
|
K
|
F
|
K
|
F
|
| Tissue type** |
Kid.
|
Kid.
|
LV
|
LV
|
Lung
|
Lung
|
Liver
|
Liver
|
SC
|
SC
|
| Wt. of tissue**** |
0.57
|
1.24
|
0.275
|
0.50
|
0.56
|
0.33
|
0.38
|
0.55
|
0.59
|
0.42
|
| ml 1° fixative |
9.4
|
14.8
|
8.1
|
8.6
|
8.3
|
7.5
|
8.2
|
13.2
|
6.9
|
6.2
|
| For first dilution (a): | ||||||||||
| VPFR (ml) |
0
|
5
|
0
|
0
|
0
|
0
|
0
|
3
|
0
|
0
|
| VDA (ml) |
9.9
|
10.8
|
8.3
|
9
|
8.7
|
7.8
|
8.5
|
10.6
|
7.4
|
6.5
|
| FVAFD (ml) |
19.3
|
20.6
|
16.6
|
17.6
|
17.0
|
15.3
|
16.7
|
20.8
|
14.3
|
12.7
|
| For second dilution, one-half of the FVAFD was replaced with fresh diluent. For final dilution and details of osmication, see text. * 1° = primary; K = Karnovsky’s fixative; F = formalin fixative. |
It was assumed that each tissue sample was impregnated with roughly 3 molar glycerol, which should be washed out gradually to avoid any significant possibility of osmotic distortion of the tissue. The first dilution employed was intended to reduce glycerol concentration to one half of the initial value. However, this initial value was not known with certainty, nor was it certain that all tissues contained the same initial concentration of glycerol. Consequently, it was decided to dilute all samples by precisely the same factor rather than diluting them to precisely the same final concentration. Millonig’s buffered formaldehyde fixative (MBFF, modified from the original formula; composition given in Table 2) was therefore added to the original fixatives in measured volumes, taking into account both the volume of original fixative present and the approximate volume of tissue water/glycerol present (assumed to be 80% of tissue weight). In cases for which the test tubes could not accept the required volume of MBFF, a measured volume of original fixative was discarded and the amount of MBFF needed was corrected and added. The exact details are given in Table 1. After the first dilution step, the samples were stored near 0°C and inverted several times on March 22 to ensure thorough mixing. The second dilution step was carried out on March 27. This time the dilution was performed by discarding one-half of the volume of fixative present in each tube and replacing it with MBFF. The third and final dilution step took place on March 30. This dilution was made by decanting the tissues and transferring them to the cryoprotectant-free solutions described in the next section.
Table 2: Composition of MB, MBFF, and Phosphate Buffer
| A. MB (Millonig’s buffer, modified) Dissolve 1.42 grams of Na2HPO4 (MW 141.96; final concentration, 99.83 mM), 0.33 grams of NaH2PO4.H2O (MW 137.99; final concentration, 24.17 mM), and 0.082 grams of sodium chloride (MW 58.44; final concentration, 14 mM) in distilled water and bring to 100 ml with distilled water. The resulting solution has an osmolality of about 294 and a pH of about 7.35. |
| B. MBFF 1. Make 100 ml of double-strength MB. 2. “Dissolve” 7.4 grams of paraformaldehyde powder in distilled water, bring to 100 ml with additional distilled water. Heat to 60-70°C. Add 2N NaOH dropwise until solution mostly clears. Cool to room temperature. Carry out step 2 in a hood using mask and gloves to avoid inhalation of powder. 3. Add 100 ml of double-strength MB to the 100 ml of 7.4% formaldehyde. Set pH to 7.4 Add concentrated CaCl2 dropwise, with stirring, until a precipitate forms. Filter the solution, label it “3.7% MBFF,” and refrigerate. Osmolality: about 1640 mOsm. |
| C. Phosphate buffer Dissolve 1.65 grams of Na2HPO4 (final concentration, 116.2 mM) and 0.33 grams of NaH2PO4.H2O (final concentration, 24.2 mM) in water, bring to 100 ml. pH = 7.4, osmolality = 304. |
Osmication and Further Processing. At the time of complete cryoprotectant washout, all tissues were separated into two additional categories, tissues to be osmicated and tissues to be processed further without osmication. Tissues not designated for osmication were decanted and placed into the 10 ml of MBFF containing no glycerol. The remaining tissues were decanted and placed into 10 ml of MB containing neither glycerol nor fixative. The latter solution was replaced the next day with 4 ml of fresh MB. The osmolality of the first Millonig’s rinse was 334 mOsm as measured at this time (March 31). Tissues designated for osmication were transferred to 9 ml aliquots of 0.5% OsO4 in isotonic sodium phosphate buffer (formula given in Table 2) on April 2. At this time, the osmolality of the second Millonig’s rinse was found to be 309 mOsm. The tissues were allowed to remain in osmium until April 8th, at which time they were rinsed with 2 ml of phosphate buffer and allowed to soak overnight in 7 ml of fresh phosphate buffer. On 4/9/84 the buffer was replaced with 4 ml of MBFF and sent to American Histolabs, Inc. (Rockville, MD) for parafin embedding, sectioning, mounting, and staining.
RESULTS
All tissues were examined both with and without osmium postfixation. However, in all cases except for that of the spinal nerves and spinal cord, the non-osmicated tissues were deemed to be the most revealing. Consequently, only osmicated tissue sections will be presented here except for the special case of spinal nerves and spinal cord. All tissues were also examined after primary fixation in either Karnovsky’s or formalin (giving 20 experimental groups examined in all: 5 tissues times two fixatives times two categories for osmication or non-osmication). However, the histological results with Karnovsky’s appeared to be identical to those obtained with formalin. Consequently, no attempt will be made below to systematically present results based on any one of these two primary fixatives. Except as noted below (for heart and spinal nerves), the original magnification was the same for all photomicrographs and was 100X before photographic enlargement.
1. LIVER. As noted above, the gross appearance of P3’s liver was extremely abnormal. The histological results bear this out. The liver consisted primarily of cavities not unlike those produced by ice as seen in freeze-substituted samples. These cavities are displayed in Figure 1. It is believe that these cavities are not, however, tell-tale signs of former ice crystals but instead are necrotic areas caused by the premortal pathology of P3. If this interpretation is correct, then these cavities reveal little about the histological consequences of freezing. Much more revealing, in this case, is the presence within the liver of “islands” of apparently well-preserved structure. Such an “island” of preserved cellular structure is shown in Figure 2. The nature of the cellular structures represented within these “oases” is not clear. What does seem clear, however, is that the degree of structural preservation is superb. The impression obtained from these areas is one of ultrastructural level/molecular level preservation, although the several processes of photographic reproduction involved in creating the image displayed in Figure 2 create a less compelling degree of clarity and “crispness” than is apparent to the naked eye during direct observation through the microscope. The degree of preservation observed is all the more impressive considering the insults suffered by P3 in addition to freezing and thawing.

Figure 1. Pathological area of P3’s liver, showing numerous cavities reminiscent of ice crystal spaces. Scale bar = 40 microns. H&E (hematoxylin and eosin stain), Karnovsky’s.

Figure 2. “Island” of well-preserved cellular structure in the liver. H&E, Karnovsky’s primary fixative. Scale bar = 40 microns.
2. LUNG. The histological structure of P3’s lung was far less affected by pathology than was the liver, and consequently appears much more normal. Two views of P3’s lung are shown in Figure 3. The lung has the typical thin-walled alveolar compartmentation pattern of normal lung (2) (A). Intact red blood cells (circled) restrained normally within apparently intact capillaries can be seen occasionally and in some lung areas (not shown) were abundant. It was also possible to observe apparently normal smooth muscle (Fig. 3B, bracketed by arrowheads), whose characteristically thin nuclei give the typical zebra-stripe look of smooth muscle. Note the crisp, intact appearance of cell nuclei in all areas of the lung (arrows). Much of the alveolar structure appeared flattened, suggesting atelectasis, but this of change would be more likely to result from the patient’s known (1) pre-mortal pathology than from freezing and thawing.

Figure 3A. Lung. For discussion, see text. Giemsa, Karnovsky’s. Magnification as in Fig. 2 (M=F2).

Figure 3B. Lung, showing normal smooth muscle (arrowheads). Giemsa, Karnovsky’s. M=F2.
3. KIDNEY. The kidney presented a variety of different and striking appearances. Figure 4 shows one type of appearance in which the renal tubular cells appear to be torn. Note, however, the intact-appearing cell nuclei in these structures (circled). The characteristic peritubular basement membrane (arrows) appears to be intact. The glomerulus (large structure just to the right of center) presents a surprisingly and impressively normal appearance and displays an intact’s Bowman’s capsule. Figure 5 shows a similar area in which the tubules are shown in longitudinal section. Not only do the cells appear to be literally torn apart, but they are separated from the basement membrane. Note also the presence of an extensive amount of unidentified ground substance (G) filling the normally empty interstitial space. The presence of this material presumably reflects pre- and/or postmortal pathology rather than any change produced by freezing and thawing. Figure 6 shows an apparently intact arteriole surrounded by torn tubules. The second type of renal appearance is shown in Figure 7. Here the tubules do not appear to be torn and thus appear more nearly normal, but their overall appearance and the presence of material in the tubular lumina suggest that they are necrotic. The cell nuclei and the tubular basement membranes, however, appear intact, as does the glomerulus. Again, ground substance fills the interstitial space. Finally, the third appearance of the kidney is shown in Figure 8. This region, from the renal medulla, shows strikingly normal and intact appearing tubules and ducts, although there is an equally striking contraction of most tubules with separation from their surrounding basement membranes.

Figure 4. Kidney. For discussion, see text. PAS (periodic acid/shiff stain), Karnovsky’s. M=F2.

Figure 5. Kidney. For discussion, see text. PAS, Karnovsky’s. M=F2.

Figure 6. Renal arteriole. Fundamentally intact structure is apparent. PAS, Karnovsky’s used as primary fixative. M=F2.

Figure 7. Kidney. For discussion, see text. PAS, Karnovsky’s. M=F2.

Figure 8. Renal medulla. For discussion, see text. H&E, formalin. Magnification as in Fig. 1 (M=F1).
4. HEART. The appearance of the left ventricle is shown in Figure 9. The muscular bundles shown represent cardiac muscle cells arranged end-to-nd and appear to be intact, though they are separated by large extracellular spaces not normally encountered in heart. At the magnification shown in Fig. 9A, and, indeed, at the resolution normally available with the light microscope, the cardiac mitochondria cannot be seen. Muscular cross-striations are also difficult to discern at this magnification, but, as indicated at higher magnification in Fig. 9B (see arrows), they can be seen frequently. These characteristic striations attest to the surprisingly good histological preservation of the tissue and the apparent absence of thaw-rigor. The muscle fibers shown in Figure 9 also appear normal when viewed in transverse section (Figure 10A). Normal blood vessels were also observed in the left ventricle (Figure 10B). In some areas, apparent separation of myoblasts along the intercalated discs was observed, but this was not a constant finding and probably represents tearing artifacts produced by osmication (not shown).

Figure 9A. Left ventricle. Myofibrils are intact but separated. H&E, formalin primary fixative. M=F2.

Figure 9B. Higher magnification taken from the center of Fig. 9A. Note the evidently normal and plentiful muscle cross-striations. H&E, formalin.
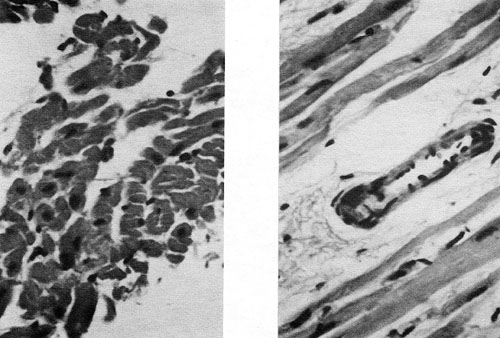
Figure 10A (left) and 10B(right). A shows cross-section of myofibrils, displaying normal shape and density. B shows a cardiac blood vessel with fundamentally preserved structure. H&E, formalin. M=F1 for both A and B.
5. SPINAL NERVES. Figure 11 displays the appearance of one of P3’s spinal nerves as shown in transverse section close to its point of origin from the spinal cord. The overall structure of the nerve in general and of the myelin sheaths in particular appears strikingly and impressively well- preserve. Unfortunately, many details visible in color are obscured in this black-and-white print and by photographic reproduction. Within the myelin sheaths (dark circular areas) shrunken but apparently intact axons can be seen, with obviously distinct boundaries (shown more clearly in the inset). It should be kept in mind that the observed shrinkage of these axons could represent a fixation artifact rather than an effect of glycerolization or of freezing and thawing. Similarly impressive preservation of another spinal nerve is shown in Figure 12. It is important to point out that these nerves are entirely representative of all such structures observed and were not selected on the basis of unusually good preservation.

Figure 11. Spinal nerve. M=F2. Inset (higher magnification) shows more detail. Osmium plus H&E (O+H&E), Karnovsky’s.

Figure 12. Second spinal nerve, showing both cross-sectional and longitudinal views of the myelinated neurons. Myelin and axons are intact. O+H&E, Karnovsky’s. M=F2.
6. SPINAL CORD. P3’s spinal cord manifested evidence of a severe undiagnosed degenerative condition primarily confined to the center of the cord. A section through this region is shown in Figure 13. As shown in Figure 13A, the central area of cord appeared to consist primarily of cavities and connective structures. However, in most cases of osmium did not penetrate to the center of the cord, and in the absence of osmium it is very difficult to see myelin. Figure 13B shows a central area of cord in which osmium did penetrate. Here we do in fact see myelin and some small myelinated fibers, but the structure of the cord is clearly degenerated. In Figure 13C, from a non-osmicated region of the center of the cord, two apparently intact, apparently nervous tissue cells can be seen. Their nature is unidentified.

Figure 13. Spinal cord. A (left): cavities in center of cord. B (center): osmicated area in center of cord, showing a few surviving axons of small diameter. C (right): two very well-preserved neurons in center of cord. A and C: H&E; B: osmium + H&E. Karnovsky’s for A-C. M=F1 for A-C.
As one proceeds from the center of the spinal cord to the periphery, one encounters a transitional zone between the clearly degenerated regions deep within the cord to strikingly well preserved regions near the cord surface. This transition zone is shown in Figure 14. Figure 15 shows an area between the transition zone and the outer edge of the cord. This region manifests excellent histological preservation, with intact myelin sheaths and intact myelinated axons, although considerable areas of non-nervous ground substance, presumably related to P3’s nervous pathology, are also present. Finally, Figure 16 shows the outer edge of the spinal cord, with typically excellent histological preservation and two apparently normal cord blood vessels which are free of blood cells (indicating that the cord in fact perfused with glycerol).

Figure 14. Spinal cord: transition zone from central to outer areas. Note apparently intact central blood vessel (V). Osmium + H&E, Karnovsky’s. M=F2.

Figure 15. Body of the peripheral part of the spinal cord. O + H&E, Karnovsky’s. M=F1.

Figure 16. Outer edge of the spinal cord. Note the two apparently intact blood vessels (V). Osmium + H&E, Karnovksy’s. M=F2.
DISCUSSION AND CONCLUSIONS
In considering the meaning of the observations reported here, we will discuss the following three general questions. 1) What can be said to have learned from the observations reported here? 2) Are our observations consistent with, and are they illuminated by, cryobiological findings obtained on simpler systems (particularly whole organs)? And finally, 3) what are the implications of our findings with respect to the repair of freezing damage in cryopreserved humans and the feasibility of cryonics in general?
1. What have we learned from the present investigation?
Caveats. With respect to this question, it is first important to define the limitations of this study. First, the observations were made on a single patient only, and could theoretically be unique to this patient, making our observations no better than “anecdotal.” Second, we do not know what the tissue levels of glycerol were in the areas subjected to investigation, so it is difficult to estimate the amount of dehydration and ice formation each of the examined tissues was subjected to and, therefore, the real resistance of the tissues to these stresses. Third, this patient was subjected to devastating premortal pathological conditions directly affecting at least two, and probably all, of the tissues examined, and this pathology, together with the considerable postmortal delay before cryopreservation, not only limited the availability of intact tissue available for examination but also could, in principle, have affected the degree of histological cryopreservation of the remaining intact tissue in either a positive or negative direction. Fourth, this report was not written by a trained histologist, microscopist, or pathologist, so we are not competent to present detailed analyses of the histological results in terms of known pathological effects or even in terms of comparison to normal tissue. Indeed, our present study lacks any control tissues for comparison. Finally, our study does not attempt to look at the ultrastructural or detailed biochemical integrity of the tissues examined and therefore provides information only on a relatively gross level of biological organization.
Results. While fully acknowledging and recognizing the above limitations, major conclusions of considerable importance can nevertheless still be drawn with confidence.
First, current methods of human cryopreservation are capable of preserving a tremendous amount of cellular and non-cellular detail even in patients suffering from extensive pathology and extensive postmortal deterioration and preserved without the desired degree of cryoprotectant permeation into the tissues. Extensive histological detail was observed in every tissue examined.
Secondly, not only the quantity but also the general quality of histological cryopreservation observed is pleasantly surprising and impressive. Apparently intact cell nuclei and blood vessels were present everywhere, seemingly ultrastructural-quality preservation was seen in intact portions of the liver, glomerular and basement membrane preservation and even tubular preservation in selected regions was observed in the kidney, distinctly intact cardiac muscle fibers with apparently normal patterns of striation were found, and the general histological organization of the lung as well as all other tissues examined was intact. Cell membranes and even capillaries appeared to escape gross structural injury as evidenced by distinctly intact red cells containing clearly visible hemoglobin and the fact that these red cells always confined to capillary lumens and were not found in the extracellular spaces surrounding the capillaries. Few clear signs of mechanical distortion and permanent alteration of the tissues by the presence of ice were found, most tissues appearing not to have been frozen at all, despite the fact that extensive freezing did in fact take place. One possible exception to this general rule was the kidney, which exhibited apparent tearing of the tubular cells in a pattern which to the authors’ knowledge is not characteristic of any known pathological condition. A perhaps more clear-cut exception was the heart, which contained large and abnormal extracellular spaces which were likely to have resulted from extracellular ice formation. On the other hand, it is also possible that these spaces simply represent cardiac edema induced by cryoprotectant perfusion.
Third, the histology of both central nervous system tissue (spinal cord) and peripheral nervous system tissue (spinal nerve) appeared to be preserved better than the histology of any other tissue. Intact nerve cell membranes and intact myelin sheaths were observed, and there was no evidence of tissue distortion by ice with the possible exception of what appeared to be microscopic fissures in the tissue. However, it is likely that these fissures are artifacts produced by the sectioning of osmicated, parafin-embedded tissue, as osmication always seems to be associated with such features even in tissues which have not bee frozen and thawed (data from a separate study). Rather extreme shrinkage of the axons within their myelin sheaths was observed. Nevertheless, the degree of preservation seen was overall, highly impressive and encouraging.
Fourth, and finally, something can be said about the biochemical state of the tissues examined. Histological stains act by chemical reacting with specific types of functional groups in the tissues. To the extent that stained tissue is observed to exhibit both the normal color and the normal intensity of stain expected for control tissue, it can be concluded that it possesses the same functional groups in the same amounts as untreated tissue. We did not find the tissue staining in this study to be discernably different from what would be expected for normal control tissue. Therefore, within very broad limits, we can conclude that extensive chemical modification of human tissue is not caused by cryopreservation of the body.
2. Relationship of current study to the cryobiological literature.
Shrinkage of axons and the spinal cord. As noted above, significant axonal shrinkage was observed histologically. One possibility is that this represents osmotic shrinkage of the axon due to permeation of glycerol into the space between the sheath and the axon without further permeation into the axon itself. Osmotic dehydration might also explain the gross shrinkage of the cord as a whole observed macroscopically (1). Although Menz’s data (3) and the data of Fahy et al. (3B) indicate that glycerol permeation into nervous tissue requires only several minutes to an hour at room temperature, permeation is apparently quite slow at 15øC or below (3C). However, if osmotic dehydration by glycerol is the cause of the observed shrinkage, it is remarkable that freezing did not alter the semipermeability of the axonal membrane to permit glycerol entry after thawing. The shrinkage may instead be a fixation artifact, as similar shrinkage has been seen in control animal material fixed without previous perfusion with cryoprotectant. In this case, shrinkage of the cord as a whole could also be fixation-related or it could be attributed to premortal pathology. A more remote possibility could be that freezing caused shrinkage of the axons and that the axons then failed to regain their normal volumes upon thawing due to being injured in some way by the freezing-thawing process. However, there is little or not precedent for this type of behavior in the cryobiological literature.
Mechanical distortion of tissues by ice. In general, little evidence for any mechanical injury to P3’s tissues could be found in this study. Although there is currently basic unanimity within the organ cryopreservation field that mechanical injury from ice is a major causative factor in the failure of presently available organ freezing procedures, the observations made here showing little or in some cases no evidence for this type of injury are in agreement with recent studies which point toward ways of avoiding mechanical damage. In particular, considerable recent research associated with the MRC Medical Cryobiology Group in Cambridge in the United Kingdom (4,5), involving both smooth muscle strips and whole rabbit kidneys, has shown that extremely slow cooling, on the order of the cooling rates used for the freezing of P3, causes ice to form in a pattern which prevents or greatly reduces the disruption of extracellular architecture of these systems which is otherwise caused by freezing at normal rates.
Even at higher rates, however, mechanical injury may not be detectable. For example, a classical study of Meryman’s, intended to evaluate the significance of mechanical distortion of tissue by ice, found that even after extreme tissue distortion by ice, as revealed by freeze substitution, thawed liver resumed an essentially normal appearance (6). It has also bee reported that the entire leg or foot of various animals can survive freezing to -15øC (or even to dry ice temperatures, according to H.T. Meryman) without the benefit of a cryoprotective agent (7) despite the massive distortion of tissue structure which must be inevitable during such extreme conditions of freezing. It has been known for years that intertidal animals such as snails, oysters, and mussels, which survive extreme conditions of freezing for months at a time, do so despite almost unbelievable mechanical distortion by ice (8), which is not apparent upon thawing. In a review of organ preservation published many years ago, Robertson and Jacob called the histological appearance of tissues after freezing and thawing “unremarkable” (9). Slow freezing of at least 80-90% of the water in isolated canine lungs in the absence of cryoprotection was found to be compatible with survival of the lungs, as determined by acceptable function after transplantation in many cases and by good histological preservation (10), in agreement with the present results.
In our experience, the appearance of P3’s cardiac muscle, which contained exaggerated extracellular spaces presumably produced by the former presence of extracellular ice, is virtually identical to the appearance of frozen-thawed rabbit skeletal muscle as reported by Meryman (7). On the other hand, P3’s heart muscle also appears extremely similar to the control hearts of Lillehei et al. (11). Mechanical distortion of the skeletal muscle of human bodies frozen without a cryoprotectant (12) is apparently more severe than what we observed.
The apparent tearing of renal tubular cells reported seems to be without precedent. It seems unlikely to be due to mechanical effects of ice formation. Toldeo-Pereyra, for example, did not observe this type of injury in human kidneys frozen very quickly to nearly the glass transition temperature (13), and tearing similarly appears absent in kidneys frozen very slowly to dry ice temperature (5). It is possible that this injury is a form of microscopic fracturing which takes place only below the glass transition temperature to relieve long-range thermal stresses which cause other organs to fracture macroscopically but which do not in general produce macroscopic fractures of the kidney (1). But this interpretation is at odds with the observations of others (14,15), who did not observe cell tearing even though they froze kidneys to below the glass transition temperature. Perhaps this pattern was somehow produced as a result of premortal pathology, e.g., as a result of the dense ground substance which could have prevented normal thermal contraction of tubules, or perhaps it is an artifact of some kind. Further research will be necessary to elucidate the cause of this unusual type of injury. On the other hand, we found no evidence for histological injury to the renal glomeruli. This type of injury is known to be greatly diminished or prevented at the cooling rates experienced by P3 (5).
We also found no evidence of unravelling and disruption of myelin sheaths as reported by Menz (3) in his study of the freezing of cutaneous nerves, which could be a mechanical effect of freezing. This disagreement is almost certainly because Menz’s nerves were frozen abruptly in the absence of cryoprotectant while P3’s nervous system was frozen very slowly in the presence of glycerol. Menz’s study also showed that dimethyl sulfoxide may cause focal unravelling of myelin similar to that seen in rabbit brains perfused with dimethyl sulfoxide-containing solutions (3C). A very recent study of Jensen et al. (15B) also reported fragility of frozen-thawed rat hippocampal grafts frozen with dimethyl sulfoxide, further suggesting a problem with this cryoprotectant for brain. However, Menz found no such effect of glycerol, in agreement with P3’s histological picture.
It seems clear that the findings in this study are in reasonable general agreement with, and are compatible with, other findings in cryobiology concerning mechanical effects of ice (and the lack thereof) on tissue histology.
Thaw rigor. One possible point of disagreement, however, was our failure to observe “thaw rigor,” which is rigor mortis produced by freezing and thawing (16). The absence of thaw rigor was inferred from the seemingly normal striation pattern observed (1). It is not clear why we failed to observe the histological pattern of thaw rigor. One possibility is that, in view of the long postmortem delay preceding cryopreservation, the cardiac muscle passed through both rigor mortis and secondary relaxation prior to cryopreservation. Love (16) has noted that very slow freezing prevents thaw rigor by allowing for depletion of ATP during cooling, which is therefore not available to cause rigor upon thawing.
Chemical effects of freezing. Normal histological staining and normal histochemical reactions after the freezing and thawing of kidneys have been reported by others (14). It is also clear that chemical changes produced by freezing must in general be rather limited, or it would not be possible for most cells to survive freezing or for analyses to be made of a nearly infinite variety of biochemical constituents of cells after freezing and thawing, as is commonly done. Our results simply confirm for cryopreserved human bodies, in a limited way, what is already known to be true for the great majority of other systems.
3. Implications
The feasibility of reversal of cryopreservation injury. The present results do have an important bearing on the question of repair. The following conclusions seem clear.
First, despite examples in which the observed degree of injury is severe, particularly in the case of the kidney, wherein there appeared to be a physical disruption of the tubular cells, there is never any doubt as to the identity of the tissue being examined. Kidney is obviously kidney, lung is obviously lung, and so on. (Due to the patient’s pathology, we are unable to comment definitively on the liver, but it is likely also to follow the same pattern, particularly in view of Meryman’s results cited above.) Furthermore, the normal biochemical nature of these easily identifiable tissues appears to be largely unaltered. It follows from these facts that cellular (17) or molecular (18,19) repair machines, if they can be made at all, will have no trouble identifying their environment and proceeding to make appropriate repairs. The observations suggest that the amount of molecular repair required should not be large compared to the overall molecular inventory of the tissue, and therefore that the degree of molecular repair required should fall comfortably within the range of repair capability thought to be possible (18,19). On the other hand, it must be acknowledged that it is not easy to visualize how molecular machines would be able to repair large-scale structural flaws such as those seen in the kidney (torn cells) or those seen on a more gross level as macroscopic fractures (1).
Second, regardless of how much injury was present in a given tissue, even in the case of the liver (in which almost no discernable cell structure at all was present other than what was found in the small “islands”), it was always found that cell nuclei were intact and easy to identify. It follows that the genetic information necessary to identify a given cell and therefore to repair a given tissue will probably be available in practically every cell in the body, despite prolonged periods of postmortal deterioration. This conclusion is supported by the apparent stability of the genome in the face of either freezing (20) or postmortem deterioration (21). Even if DNA is significantly degraded within 30-60 min. of death (22,23), the resulting fragments should still provide ample information for cellular or molecular repair devices to decipher and act upon particularly as these fragments will all presumably be localized within the nucleus.
Third, there was no evidence of catastrophic vascular injury in any of the sections examined. Presumably, then, if macroscopic fracturing (1) can be prevented, the vascular system should in principle be available as a delivery route for both cellular and molecular repair device.
Finally, the tissue of greatest importance, central nervous system and peripheral nervous system tissue, appears excellently preserved even under the conditions experienced by P3. This observation together with recent studies of the cryopreservation of brains suggests that, in some ways, repair of the brain may be even simpler than repair of the remainder of the body. Naturally, however, much more information on the status of the brain and of the body after cryopreservation is still urgently required. Electron microscopic results obtained on a dog frozen using dimethyl sulfoxide, reported by Gale (24), indicate more CNS damage than is hinted at by the present results with P3.
Overall, this initial study shows that it is feasible to preserve histological detail in humans by cryopreservation after death. Since damage is likely to be more structural than chemical and since this study shows significant structural preservation can be achieved, the results are consistent with general feasibility of Ettinger’s proposal for the rescue of contemporary people suffering from incurable terminal diseases (25). Of course, only many, many years of decades of additional research will be sufficient to establish or rule out the true feasibility of this approach.
REFERENCES
1. Federowicz, M., Hixon, H., and Leaf, J.D., Postmortem examination of three cryonic suspension patients, Cryonics, 9/84 (#50), 16-28, 1984.
2. Bloom, W., and Fawcett, D.W., A Textbook of Histology, Ninth Edition W.B. Saunders Co., Philadelphia, 1968.
3. Menz, L.J., Structural changes and impairment of function associated with freezing and thawing in muscle, nerve, and leucocytes. Cryobiology, 8, 1-13, 1971. 3B. Fahy, G.M., Takahashi, T., and Crane, A.M., Histological cryoprotection of rat and rabbit brains, Cryo-Letters, 5, 33-46, 1984.
4. Taylor, M.J., and Pegg, D.E., The effect of ice formation on the function of smooth muscle tissue stored at -21 or -60øC, Cryobiology, 20, 36-40, 1983.
5. Hunt, C.J., Studies on cellular structure and ice location in frozen organs and tissues: the use of freeze-substitution and related techniques, Cryobiology, 21, 385-402, 1984.
6. Meryman, H.T., Ice Crystal Formation in Frozen Tissues. Lecture and Review Series, Naval Medical Research Institute, No. 53-3, 25-48,1953.
7. Meryman, H.T., “Review of Biological Freezing,” in Cryobiology (H.T. Meryman, ed.), Academic Press, New York, 1-114, 1966, and unpublished observations.
8. Kanwisher, J., Histology and metabolism of frozen intertidal animals, Biol. Bull., 116, 258-264, 1959.
9. Robertson, R.D., and Jacob, S.W., “The Preservation of Intact Organs,” in Advances in Surgery, vol. 3 (C.E. Welch, ed.), Year Book Medical Publishers, Chicago, 1968.
10. Okaniwa, G., et al., Studies on the preservation of canine lung at subzero temperatures. J. Thorac. Cardiovasc. Surg., 65, 180-186, 1973.
11. Lillehei, R.C., et al., In vitro preservation of whole organs by hypothermia and hyperbaric oxygenation, Cryobiology, 1, 181-193, 1964.
12. Reuter, K., Dtsch. Z. ges. gerichtl. Med., 1, 330, 1922; quoted in Love (ref below) (not checked)
13. Toledo-Pereyra, L.H., and MacKenzie, G.H., Freezing of human kidneys, initial in vitro observations, The American Surgeon, 48, 232-236, 1982.
14. Schimmel, H., Wajcner, G., Chatelain, C., and LeGrain, M., Freezing of whole rate and dog kidney by perfusion of liquid nitrogen through the renal Surg. Forum, 9, 802-804, 1958.
15B. Jensen, S., Sorenson, T., Moller, A.G., and Zimmer, J., Intraocular grafts of fresh and freeze-stored rat hippocampal tissue: a comparison of survivability and histological and connective organization, J. Comp. Neurol., 227, 558-568, 1984.
16. Love, R.M., “The Freezing of Animal Tissue,” in Cryobiology (H.T. Meryman, ed.), Academic Press, New York, pp. 317-405, 1966.
17. Darwin, M.g., The anabolocyte: a biological approach to repairing cryoinjury, Long Life Magazine, 1, 80-83, 1977.
18. Drexler, E., The Future of Design, to be published.
19. Drexler, E., Cell Repair Machines and Tissue Reconstruction: Some Notes on Computational Complexity and Physical Constraints, to be published.
20. Elliott, K., and Whelan, J., eds., The Freezing of Mammalian Embryos, Elsevier/Excerpta Medica, New York, 1977.
21. Gilbert, J.M., et al., The preparation of biologically active messenger RNA from human postmortem brain tissue, J. Neurochem., 36, 976- 984, 1981. This reference is actually to mRNA, not DNA. However, the hardiness of RNA suggest hardiness of DNA. In addition, the intact mRNA could be transcribed into DNA using reverse transcriptase (this is well- established technique).
22. Lazarus, H.M., and Hopfenbeck, A., DNA degradation during organ storage, Experientia, 30, 1410-1411, 1974.
23. WIlliams, J.R., Little, J.B., and Shipley, W.U., Association of mammalian cell death with a specific endonucleolytic degradation of DNA, Nature (Long.), 252, 754-755, 1974.
24. Gale, L., Alcor experiment: surviving the cold. Long Life Magazine, 2, 58-60, 1978.
25. Ettinger, R.C.W., The Prospect of Immortality, Doubleday, 1964.
